
Rutherford’s planetary model of the atom was based upon classical physics – a system that deals with physical particles, force, and momentum. While Rutherford’s model explained many observations accurately, it was found to have flaws. Like so many before him, Bohr built upon the work of his predecessors, and for Bohr, part of that foundation had been built by Ernest Rutherford.īased upon a series of experiments, Rutherford proposed the planetary model of the atom in which electrons swirled around a hard, dense nucleus (see Atomic Theory I: The Early Days). 
The most intense period of progress took place between the late 19 th and early 20 th century, and it hinged heavily on the work of a Danish physicist named Niels Bohr. But that was all about to change, and quite dramatically. Given that many centuries had elapsed between the earliest ideas of the atom and Dalton’s work, it would be fair to say that the evolution of atomic theory had been a gradual one, with progression in the field being steady rather than spectacular. French chemists Antoine Lavoisier and Joseph Proust, with their Law of Conservation of Mass in 1789 and Law of Definite Proportions in 1799, respectively, each laid the groundwork for Englishman John Dalton’s work on the Law of Multiple Proportions (Dalton, 1803). The story of atomic theory first encounters reproducible, scientific (evidence based) proof in the late 18th century. In fact, one could argue that the history, struggle, and achievement that is threaded through the development of understanding matter at the atomic level is the quintessential story of the scientific method. This gradual, logical progression, where atomic structure evolved from being a simple, philosophical idea, through to the ultra-sophisticated world of the Higgs boson particle discovered in the early part of the 21 st century, represents a wonderful example of the evolution of a scientific idea, and the application of the scientific process. 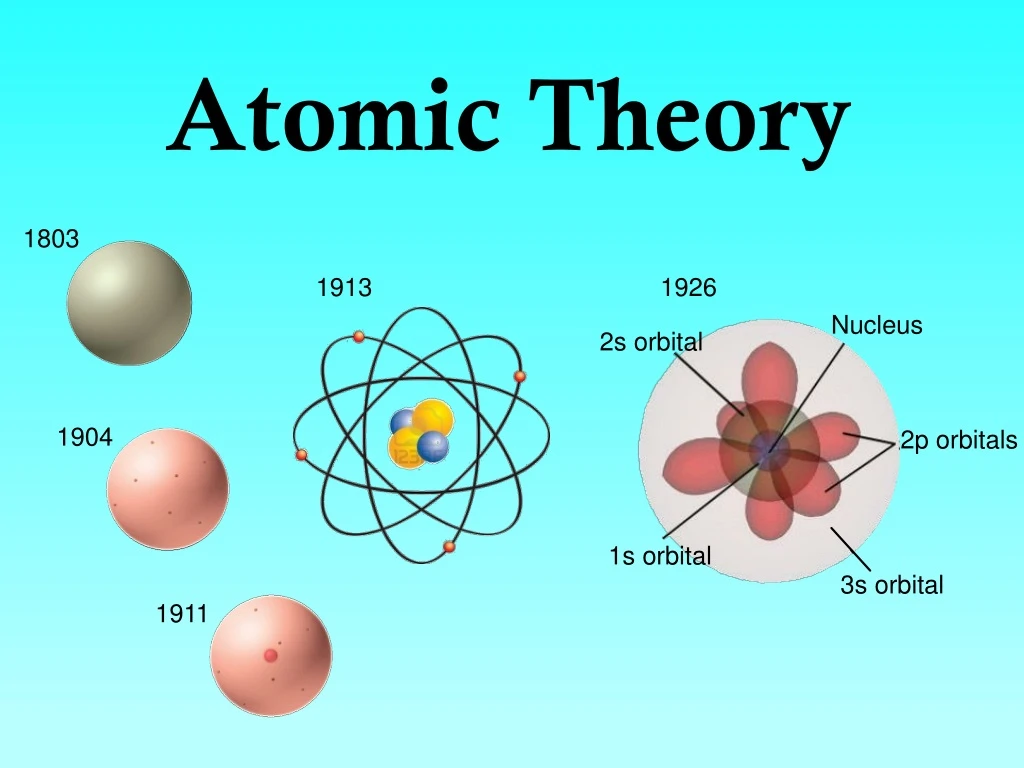
(You can read more about this is in our modules Early Ideas about Matter: From Democritus to Dalton and Atomic Theory I: The Early Days.) Despite the slow pace, it is crucial to understand that the process was a methodical one as each scientist built upon earlier ideas. Starting with the ancient Greeks, and moving through to the beginning of the 19th century, the story unfolds relatively slowly. The total mass of all products of a chemical reaction is equal to the total mass of all reactants of that reaction.The earliest ideas about matter at the atomic level were built over many centuries. 
Finally, we all assume that we have demonstrated the Law of Conservation of Mass. The elements are not transmutable: one element cannot be converted into another. For example, metallic iron and gaseous oxygen are both elements and cannot be reduced into simpler substances, but iron rust, or ferrous oxide, is a compound which can be reduced to elemental iron and oxygen. All other pure substances, which we call compounds, are made up from these elements and can be decomposed into these elements. We will assume that we have identified all of these elements, and that there are a very small number of them. We will begin by assuming that all materials are made from elements, materials which cannot be decomposed into simpler substances. There are over 18 million known substances in our world.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
